 |
|
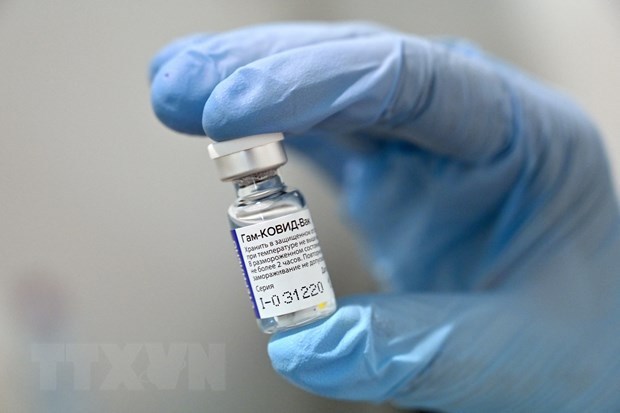
Russia’s COVID-19 vaccine Gam-COVID-Vac, also known as Sputnik V |
It is the second COVID-19 vaccine to be authorised in Vietnam.
Conditions for the authorisation of the vaccine are also promulgated.
The Drug Administration of Vietnam at the Ministry of Health is requested to be responsible for the imports of the vaccine and comply with the regulations on imported vaccine quality management.
Meanwhile, the MoH’s Administration of Science Technology and Training is to roll out clinical assessment in Vietnam on the safety and immune response of the vaccine.
Although the third phase of clinical trial has not been completed, the Russian-developed vaccine has been approved in more than 50 countries, and is 91.8 percent effective against the coronavirus SARS-CoV-2.
Of note, it produces antibodies in 98 percent in volunteers after they were given shots of the vaccine./. VNA
Ministry asked to approve US, Russian COVID-19 vaccines
Vaccination will start in early March, and 1.2 million more doses may be added to the programme in late March.